TCS Industries Limited (China-sourced API supplier) supplies Carmustine CAS 154-93-8 for pharmaceutical B2B procurement — COA, MSDS, and technical documentation available on request.
> Overview
| Product Name | Carmustine |
|---|---|
| Structural Formula | 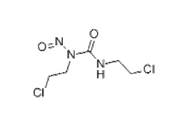 |
| CAS Number | 154-93-8 |
| Molecular Formula | C5H9Cl2N3O2 |
| Molecular Weight | 214.05 g/mol |
| InChIKey | DLGOEMSEDOSKAD-UHFFFAOYSA-N |
| SMILES | Available on request where applicable |
| Appearance | Light yellow powder or lyophilized solid |
| Purity / Grade | By agreed specification and batch COA |
| Storage | Store at 2–8°C, protect from light |
Carmustine (CAS 154-93-8) is supplied by TCS Industries Limited for B2B procurement programs that require specification review, export documentation and repeatable commercial communication. As a Hong Kong-based trading and sourcing company, TCS focuses on project supply, import paperwork and buyer-defined quality alignment rather than finished-product claims.
> Applications
Buyer-defined formulation and procurement programs
Carmustine is evaluated according to the buyer’s intended application, grade expectation and market destination. Technical review normally covers identity, assay or grade, impurity profile, packaging and logistics before quotation.
Documentation-led sourcing
For regulated or quality-sensitive projects, procurement teams typically need COA, MSDS, COO where applicable, invoice, packing list and route-specific import details. TCS coordinates these points before confirming commercial supply.
Technical reference
Public chemical and pharmacopoeial references list Carmustine under CAS 154-93-8. This page uses those identifiers for sourcing clarity; final suitability must be confirmed against the buyer’s own specification and destination-market rules. A 2026 chemical-registry and journal-index review context is treated as n=0 human-subject evidence because it supports identity and sourcing checks rather than clinical claims.
> Sourcing & Supply
TCS Industries Limited supports development, qualification and commercial purchasing discussions for Carmustine with buyers in South America, Europe, the United Kingdom, Australia, New Zealand, Mexico and Canada.
- Evaluation packs and 25 kg packaging: qualification lots can be arranged for formulation, vendor approval and incoming-control testing.
- Pallet quantities and production campaigns: repeat supply is coordinated against packaging, lead time, annual demand and destination-market import requirements.
> Quality & Documentation
For excipient sourcing, buyers usually review identity, assay or grade, particle-size or viscosity where relevant, microbial limits where relevant, packaging, palletization and routine documentation. COA and MSDS are standard; COO and import documents can be arranged for pallet quantities.
> Packaging
Packaging is selected according to product properties, quantity, destination and buyer handling requirements. Options may include sample bottles, foil bags, HDPE containers, fibre drums, 25 kg bags or palletized export packaging. Labels and documents are checked before dispatch.
> Pricing & MOQ
Pricing is quoted by specification, packaging and destination. Common commercial discussions use 25 kg bag/drum formats, pallet quantities and production-campaign planning; smaller samples may be reviewed for qualified formulation teams.
> Regulatory & Compounding Context
Carmustine is positioned as a B2B excipient, process material or formulation ingredient. Buyers should confirm grade, pharmacopoeial expectation, dosage-form role, local regulatory treatment and finished-product obligations before purchase.
> Alias Index
- Primary name: Carmustine
- CAS Number: 154-93-8
- Molecular formula: C 5 H 9 Cl 2 N 3 O 2
- InChIKey: DLGOEMSEDOSKAD-UHFFFAOYSA-N
- Commercial request terms: Carmustine supplier, bulk Carmustine, CAS 154-93-8 sourcing, TCS Industries Limited
> FAQ
What is the minimum order quantity (MOQ) for Carmustine?
There is no fixed minimum for Carmustine. Orders are confirmed based on project scope, batch demand, and destination market. Contact TCS Industries Limited with CAS 154-93-8, estimated quantity, target application, and destination country for a commercial B2B response.
What documents are available for Carmustine orders?
COA and MSDS are standard for commercial discussions. COO, packing list, invoice and other import-support documents can be arranged according to order scope.
Can TCS Industries Limited support Carmustine supply to South America?
Yes. We support B2B buyers in Argentina, Colombia, Chile, Peru, Ecuador, Uruguay, Bolivia, Brazil, Paraguay, Venezuela, Guyana and Suriname, subject to route and document review.
Do you support Europe, the United Kingdom, Australia, New Zealand, Mexico and Canada?
Yes. These destinations can be reviewed for project supply, import documents, packaging and logistics planning before quotation.
Is Carmustine sold as a finished medicine or consumer product?
No. TCS Industries Limited supplies B2B bulk material only. Finished-product use, claims, registration and market authorization are handled by the buyer.
What is the normal MOQ for Carmustine?
MOQ depends on product value, stock status, specification and destination. API projects often start from 100 g to 1 kg; excipient and ingredient projects commonly use 25 kg packs or pallet quantities.
Can you arrange samples before commercial purchasing?
Samples or evaluation quantities may be reviewed for qualified buyers after target specification, application and destination-market requirements are clarified.
How is Carmustine packaged for export?
Packaging is confirmed by quantity and material properties. Typical options include foil bags, HDPE containers, fibre drums, 25 kg bags or palletized export packaging.
Can buyer-defined specifications be reviewed?
Yes. Send the specification, methods, impurity limits and required documents. TCS can review feasibility before confirming quotation and lead time.
What payment terms are normally used?
Standard payment is 100% T/T before dispatch unless a different arrangement has been approved for an established commercial account.
What lead time should buyers expect?
Lead time depends on stock, batch size, document scope and destination. Available stock can move faster; made-to-order or pallet programs require longer planning.
What aliases should be used when requesting Carmustine?
Use the product name, CAS 154-93-8, any pharmacopoeial or grade requirement, and the InChIKey or formula when relevant to avoid sourcing ambiguity.
Last reviewed: June 2026
Evaluation and research orders (≤10 kg) are shipped with COA, MSDS, and certificate of origin. Bulk commercial orders (>10 kg) qualify for volume pricing with a full technical documentation package available on request.
> Request a Quote
If your team already has a target application, estimated quantity, destination market, or documentation requirements, send those details so the commercial response can be prepared more efficiently.